Selecting the correct internal coating is not only about chemical resistance. In many cases, improper coating choice is a direct cause of leakage, corrosion, and shipment failure.
If leakage prevention is your priority, you may also find this guide helpful: Prevent Chemical Tin Can Leaks: A Systems Approach
Struggling with leaking cans and product contamination? A failed coating can ruin your product and your reputation. Following a professional decision framework ensures your chemical products are safe and stable.
To choose the right internal coating for a chemical can, you must prioritize chemical compatibility1 over all other factors. This means identifying the product's chemical properties (pH, polarity, reactivity) before considering coating material, cost, or can manufacturing process. This chemistry-first approach prevents most corrosion and leakage issues2.

Choosing the right internal lining for a chemical can feels like a complex technical challenge, but it doesn't have to be. Over my nearly two decades in this industry, I've seen it all. The good news is that most failures can be avoided by following a clear, logical process. It's not about being a chemist yourself, but about asking the right questions. Let's walk through the exact framework we use to ensure our customers' products are packaged safely and effectively, every single time.
Choosing the right internal lining for a chemical can feels like a complex technical challenge, but it doesn't have to be. Over my nearly two decades in this industry, I've seen it all. The good news is that most failures can be avoided by following a clear, logical process. It's not about being a chemist yourself, but about asking the right questions. Let's walk through the exact framework we use to ensure our customers' products are packaged safely and effectively, every single time.
What Is the Biggest Mistake When Choosing Internal Coatings?
You've used the same type of can for years, so you stick with it. Then, a new product formulation causes unexpected leaks and customer complaints. The problem isn't the can; it's the process.
The biggest mistake is selecting coatings by habit instead of by chemical compatibility. A coating failure often happens not because the coating itself is "low quality," but because the specific chemical properties of the contents were misunderstood or simply not considered.

In my experience, this is the number one cause of packaging failure. I remember a client who produced a line of industrial cleaners. They always used a standard epoxy-phenolic lined can3, and it worked perfectly for years. Then, they launched a new "green" formula. They assumed the same can would be fine—after all, the new product was less harsh. Six months later, we got an urgent call about widespread leakage. The new formula had a slightly different pH that slowly attacked the coating, creating tiny pinholes. This taught us a powerful lesson: in chemical packaging, coating selection is a chemistry problem, not a purchasing problem. A purchasing manager sees a can and a price. A packaging expert must see a complete chemical containment system4, where the interaction between the product and the lining is the most critical factor for success.
In my experience, this is the number one cause of packaging failure. I remember a client who produced a line of industrial cleaners. They always used a standard epoxy-phenolic lined can3, and it worked perfectly for years. Then, they launched a new "green" formula. They assumed the same can would be fine—after all, the new product was less harsh. Six months later, we got an urgent call about widespread leakage. The new formula had a slightly different pH that slowly attacked the coating, creating tiny pinholes. This taught us a powerful lesson: in chemical packaging, coating selection is a chemistry problem, not a purchasing problem. A purchasing manager sees a can and a price. A packaging expert must see a complete chemical containment system4, where the interaction between the product and the lining is the most critical factor for success.
What Information Must Be Confirmed Before Any Coating Decision?
You're on a tight deadline and need to order cans immediately. But ordering without key data is a huge gamble that can lead to product loss and safety risks. Get these critical facts first.
Before you even think about coating materials or costs, you must confirm the chemical's properties. This includes its chemical reactivity5 (is it oxidizing or reducing?), its pH range, its solvent polarity, and the presence of any aggressive ions6 like chlorides or sulfides.

Think of this step like a doctor diagnosing a patient. You wouldn't want a doctor to prescribe medicine without first understanding your symptoms, allergies, and medical history. In the same way, we cannot "prescribe" a coating without a full profile of the chemical it will hold. Skipping this diagnostic step is the root cause of nearly every leakage and corrosion issue I've ever investigated. For example, knowing the pH tells us if we're fighting an acid or an alkali. Knowing the solvent polarity tells us if the product will try to dissolve the coating. And identifying aggressive ions, like chloride (Cl⁻), is crucial because they are notorious for causing localized pitting corrosion, especially at weak points like weld seams. We also need to know the planned storage time and transport conditions, as time and temperature can accelerate negative reactions.
Think of this step like a doctor diagnosing a patient. You wouldn't want a doctor to prescribe medicine without first understanding your symptoms, allergies, and medical history. In the same way, we cannot "prescribe" a coating without a full profile of the chemical it will hold. Skipping this diagnostic step is the root cause of nearly every leakage and corrosion issue I've ever investigated. For example, knowing the pH tells us if we're fighting an acid or an alkali. Knowing the solvent polarity tells us if the product will try to dissolve the coating. And identifying aggressive ions, like chloride (Cl⁻), is crucial because they are notorious for causing localized pitting corrosion, especially at weak points like weld seams. We also need to know the planned storage time and transport conditions, as time and temperature can accelerate negative reactions.
Are You Packaging Strong Oxidizers, Concentrated Acids, Alkalis, or Specialty Chemicals?
You handle high-value or hazardous materials. A container failure isn't just an inconvenience; it's a potential safety disaster that could have serious consequences. You need a coating built for the toughest jobs.
If the answer is yes, then a PVF (fluorocarbon) coating7 should be your first and primary consideration. PVF offers extremely high chemical inertness, providing excellent resistance to the most aggressive acids, alkalis, and oxidizers with minimal risk of interaction.

When we're dealing with high-risk chemicals, there is no room for error. This is where we bring in the top-tier materials. PVF, or Polyvinylidene Fluoride, is a high-performance fluoropolymer. You can think of it as the ultimate defense for can interiors. Its molecular structure is incredibly stable, which makes it almost completely inert. It simply doesn't react with the vast majority of chemicals. We specify it for products like potent agricultural chemicals, high-purity solvents, and aggressive industrial agents. It's also the best choice for products that will be exported or stored for long periods. The temperature swings and physical stress of a long journey can accelerate any weakness in a standard coating. While PVF is a more premium option, it's not a cost—it's an insurance policy. For high-risk applications, over-engineering for safety is always a smarter business decision than saving a few cents on the can.
When we're dealing with high-risk chemicals, there is no room for error. This is where we bring in the top-tier materials. PVF, or Polyvinylidene Fluoride, is a high-performance fluoropolymer. You can think of it as the ultimate defense for can interiors. Its molecular structure is incredibly stable, which makes it almost completely inert. It simply doesn't react with the vast majority of chemicals. We specify it for products like potent agricultural chemicals, high-purity solvents, and aggressive industrial agents. It's also the best choice for products that will be exported or stored for long periods. The temperature swings and physical stress of a long journey can accelerate any weakness in a standard coating. While PVF is a more premium option, it's not a cost—it's an insurance policy. For high-risk applications, over-engineering for safety is always a smarter business decision than saving a few cents on the can.
If Not High-Risk Chemicals, What Is the Main Chemical Base?
Your product isn't an extreme acid or solvent, but that doesn't mean any can will do. Choosing the wrong lining can still lead to problems. The first step is to classify your product simply.
The most important question to ask next is: is your product water-based or solvent-based? This simple classification is the fork in the road that determines your entire coating strategy. It tells you whether corrosion resistance8 or solvent resistance9 is the main priority.
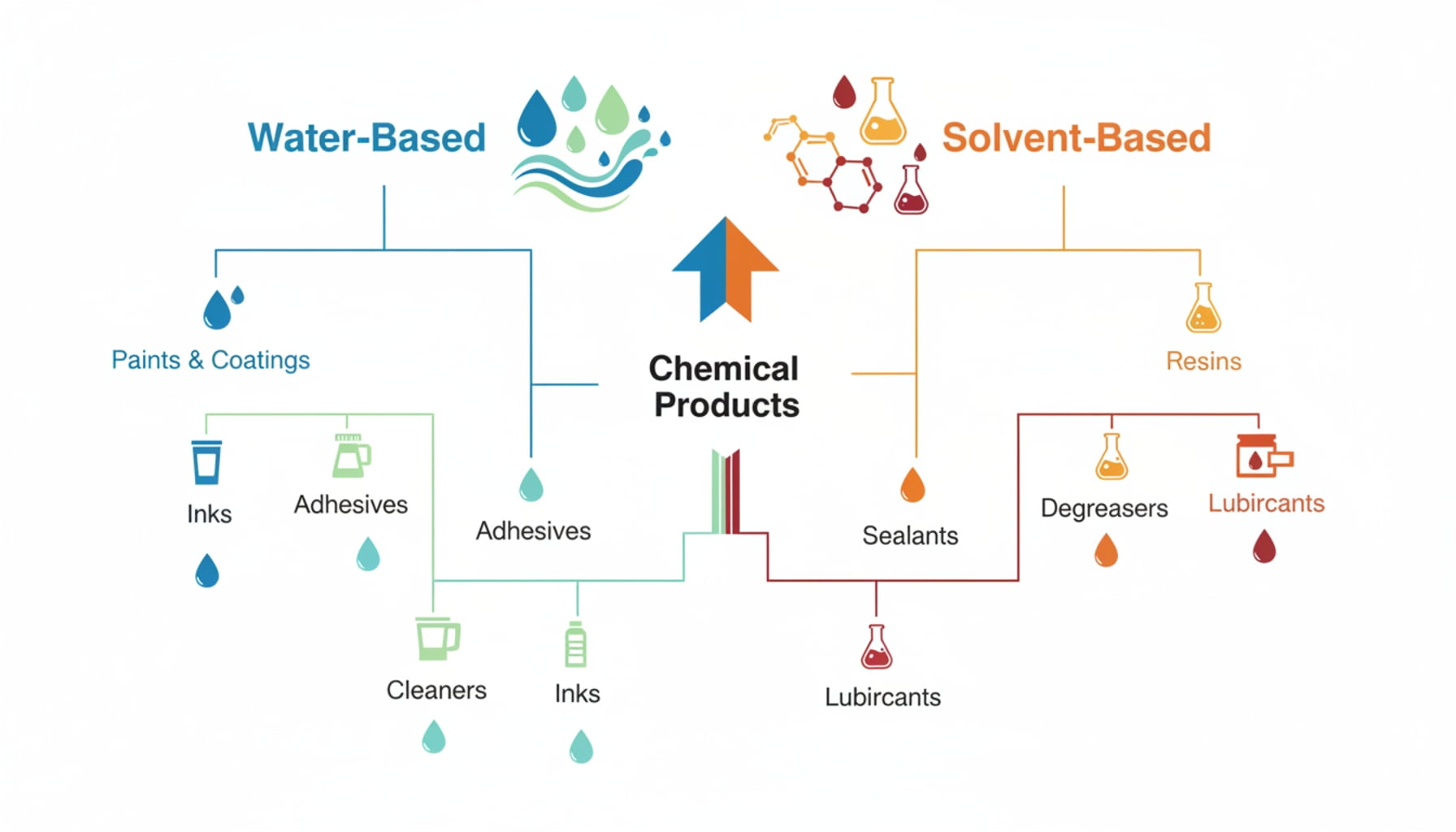
Once we've determined that a specialized coating like PVF isn't necessary, this is the very next question I ask every client. The answer immediately tells us what kind of battle we need to fight. If the product is water-based, our main enemy is corrosion. Water, especially if it contains salts or other electrolytes, will actively try to react with the steel of the can. The coating's job is to act as a perfect, impenetrable barrier to prevent this. On the other hand, if the product is solvent-based (or oil-based), our enemy is dissolution. The solvent itself won't cause the steel to rust, but it can soften, swell, or completely dissolve an incompatible coating. This can lead to contamination of your product and eventual failure of the lining. The failure mechanisms are completely different, so the solutions must be too. This fundamental classification guides all subsequent decisions.
Once we've determined that a specialized coating like PVF isn't necessary, this is the very next question I ask every client. The answer immediately tells us what kind of battle we need to fight. If the product is water-based, our main enemy is corrosion. Water, especially if it contains salts or other electrolytes, will actively try to react with the steel of the can. The coating's job is to act as a perfect, impenetrable barrier to prevent this. On the other hand, if the product is solvent-based (or oil-based), our enemy is dissolution. The solvent itself won't cause the steel to rust, but it can soften, swell, or completely dissolve an incompatible coating. This can lead to contamination of your product and eventual failure of the lining. The failure mechanisms are completely different, so the solutions must be too. This fundamental classification guides all subsequent decisions.
How Should Water-Based or Electrolyte Solutions Be Coated?
You might think "water-based" means harmless and easy to package. But add some salts or set the pH wrong, and that water can become more corrosive than some acids.
For water-based chemicals, your coating choice must focus on corrosion resistance. The selection is driven primarily by the product's pH level10 and the presence of any corrosive ions. Water-based electrolytes can be surprisingly aggressive in accelerating metal corrosion.

Many people get this wrong. They hear "water-based" and relax. In my experience, these products can be some of the most challenging to package correctly. Water is a powerful solvent, and when you dissolve salts or other electrolytes in it, you essentially create a small battery inside the can that relentlessly attacks the metal. This is why a detailed look at the formula is so important. We use a structured approach to select the right barrier.
Many people get this wrong. They hear "water-based" and relax. In my experience, these products can be some of the most challenging to package correctly. Water is a powerful solvent, and when you dissolve salts or other electrolytes in it, you essentially create a small battery inside the can that relentlessly attacks the metal. This is why a detailed look at the formula is so important. We use a structured approach to select the right barrier.
Coating Recommendations for Water-Based Chemicals
| Condition | Risk Focus | Recommended Coating |
|---|---|---|
| Strong acidity (low pH) | Metal dissolution | Epoxy / Epoxy-Phenolic |
| Strong alkalinity (high pH) | Alkaline attack on coating | High-performance Epoxy |
| Extreme alkalinity | Severe coating breakdown | PVF fluorocarbon |
| Contains sulfur/chloride ions | Pitting corrosion | Upgraded Epoxy or PVF |
Critical Note
A critical detail I always point out is that aggressive ions such as chloride and sulfur often attack the weld seam first, rather than the flat surfaces of the can.
The weld seam is a point of high stress and is therefore more susceptible to corrosion. Ensuring complete and uniform coating protection at the weld seam is critical for long-term performance.
How Should Oil-Based or Organic Solvent Products Be Coated?
Your product is an oil or an organic solvent, so you know it won't rust the can. But it might just "eat" the internal lining for lunch, causing contamination and leaks.
The survival of the coating depends on the solvent's polarity. Oil-based systems don't typically corrode the steel, but they can cause an incompatible coating to swell, soften, or dissolve entirely. The key is to match the coating's resistance to the solvent's chemical nature.
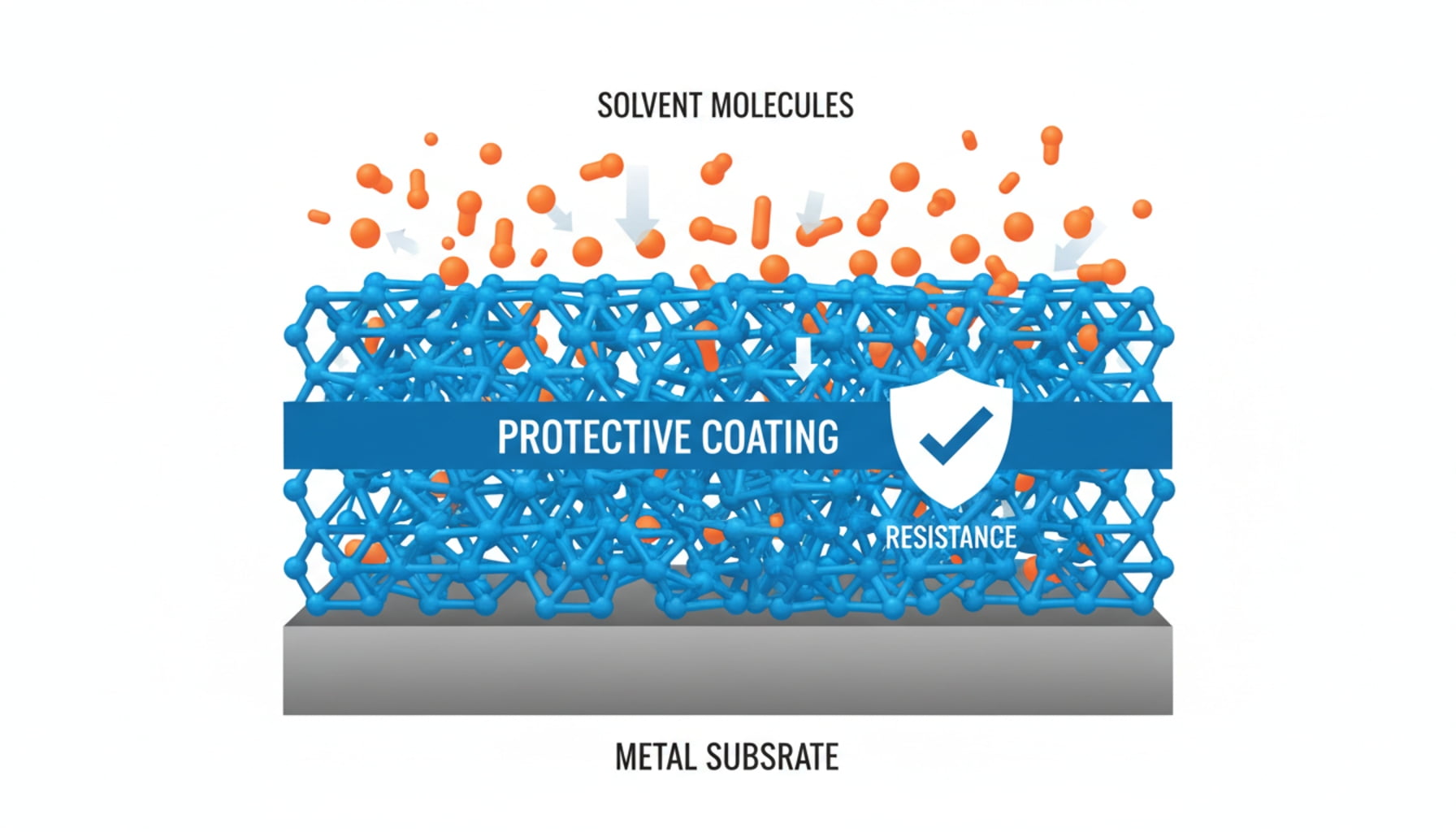
With solvent-based products, our concern shifts from preventing rust to preventing the coating itself from breaking down. A solvent’s primary job is to dissolve substances, and it will quickly attack the wrong type of lining.
With solvent-based products, our concern shifts from preventing rust to preventing the coating itself from breaking down. A solvent’s primary job is to dissolve substances, and it will quickly attack the wrong type of lining.
The guiding principle here comes from basic chemistry: “like dissolves like.” This is why understanding the solvent’s polarity is critical. Polar solvents, such as ketones and alcohols, have molecular structures that can aggressively attack many standard coatings. In these cases, epoxy-phenolic resins perform exceptionally well, as their tightly cross-linked structure provides strong chemical resistance.
For non-polar solvents, such as simple alkanes or aromatic compounds, alternative systems like phenolic resins can also offer excellent performance.
For a general overview of solvent polarity and selection principles, see:
Factors for Choosing the Right Solvents for Coatings
Solvent-Based Coating Selection Table
| Solvent Type | Typical Examples | Recommended Coating |
|---|---|---|
| Polar solvents | Ketones, esters, alcohols | Epoxy-Phenolic |
| Non-polar solvents | Alkanes, aromatics | Phenolic / Epoxy-Phenolic |
| Complex formulations | Blends with additives | Compatibility testing required |
Key Insight
The real challenge comes with modern, complex formulations that are a cocktail of different solvents and additives. In these cases, theory alone is not enough.
There is no substitute for testing the actual product against the recommended coating to ensure long-term compatibility and performance.
Does Can Manufacturing Process Affect Coating Choice?
You did your homework and chose the perfect coating based on the chemical. But after production, you find the lining is cracked and failing. What went wrong?
Yes, the manufacturing process absolutely affects the coating choice. A coating must have mechanical flexibility11 in addition to chemical resistance, especially if the can requires deep drawing12, stretch forming, or has complex geometry. The stress of forming can destroy a chemically "correct" coating.

This is a crucial detail that often gets missed. A coating might be perfect on a flat sheet of tinplate, but that same coating can crack and fail when the metal is stretched, bent, and formed into a can. If we are making a simple three-piece can with straight sides, the mechanical stress on the coating is relatively low. However, for many modern can designs, we use a process called deep drawing, where a punch forces the metal into a die to create a seamless can body. This process significantly stretches the coating. If the coating is too brittle, it will develop micro-cracks, creating invisible pathways for the chemical to reach the steel. This is why we often use more flexible epoxy or organosol coatings13 for deep-drawn cans.
This is a crucial detail that often gets missed. A coating might be perfect on a flat sheet of tinplate, but that same coating can crack and fail when the metal is stretched, bent, and formed into a can. If we are making a simple three-piece can with straight sides, the mechanical stress on the coating is relatively low. However, for many modern can designs, we use a process called deep drawing, where a punch forces the metal into a die to create a seamless can body. This process significantly stretches the coating. If the coating is too brittle, it will develop micro-cracks, creating invisible pathways for the chemical to reach the steel. This is why we often use more flexible epoxy or organosol coatings13 for deep-drawn cans.
Recommended Options for Forming Stress
| Process Requirement | Suitable Coating |
|---|---|
| Deep drawing | Flexible Epoxy |
| High deformation | Organosol |
| Welded seams | Powder-coated epoxy strip |
Additional Consideration
We always pay special attention to the weld seam on three-piece cans. The intense heat generated during the welding process can damage the internal lining in this area.
To address this risk, a special protective strip of powder-coated epoxy is applied over the weld seam, ensuring a complete, continuous, and unbroken barrier for long-term protection.
Is Compatibility Testing Really Mandatory?
You're in a hurry to get your product to market and the theoretical data looks good. Skipping a final test could save a few weeks, but it could also lead to a massive product recall.
Yes, compatibility testing14 is absolutely mandatory. No coating selection process is complete without real-world validation. Theory and data sheets are excellent guides, but only a physical test with your actual product can confirm long-term stability and safety.

I cannot stress this point enough. In all my years of experience, I have seen cases where a coating should have worked perfectly on paper, but a minor, unlisted trace ingredient in the customer's formula caused a slow reaction that only became apparent after months on the shelf. This is precisely why we insist on thorough compatibility testing14 as the final step. It's a non-negotiable part of our professional process. We take the recommended can and fill it with the customer's exact product. Then, we perform a series of tests, including long-term immersion and accelerated aging at elevated temperatures to simulate a long shelf life in a short period. We look for any sign of trouble: coating blisters, softening, discoloration, or any corrosion on the metal. This might add a little time to the project upfront, but it prevents catastrophic and costly failures down the road.
I cannot stress this point enough. In all my years of experience, I have seen cases where a coating should have worked perfectly on paper, but a minor, unlisted trace ingredient in the customer's formula caused a slow reaction that only became apparent after months on the shelf. This is precisely why we insist on thorough compatibility testing14 as the final step. It's a non-negotiable part of our professional process. We take the recommended can and fill it with the customer's exact product. Then, we perform a series of tests, including long-term immersion and accelerated aging at elevated temperatures to simulate a long shelf life in a short period. We look for any sign of trouble: coating blisters, softening, discoloration, or any corrosion on the metal. This might add a little time to the project upfront, but it prevents catastrophic and costly failures down the road.
How Do Professionals Make the Final Coating Decision?
This all seems very complex and it's easy to get lost in the details. You just want a reliable can, but you're worried about making the wrong choice.
Professionals remove the guesswork by following a structured decision logic15. Instead of making assumptions, we use a step-by-step framework that starts with chemistry and ends with physical testing, ensuring every variable is accounted for.
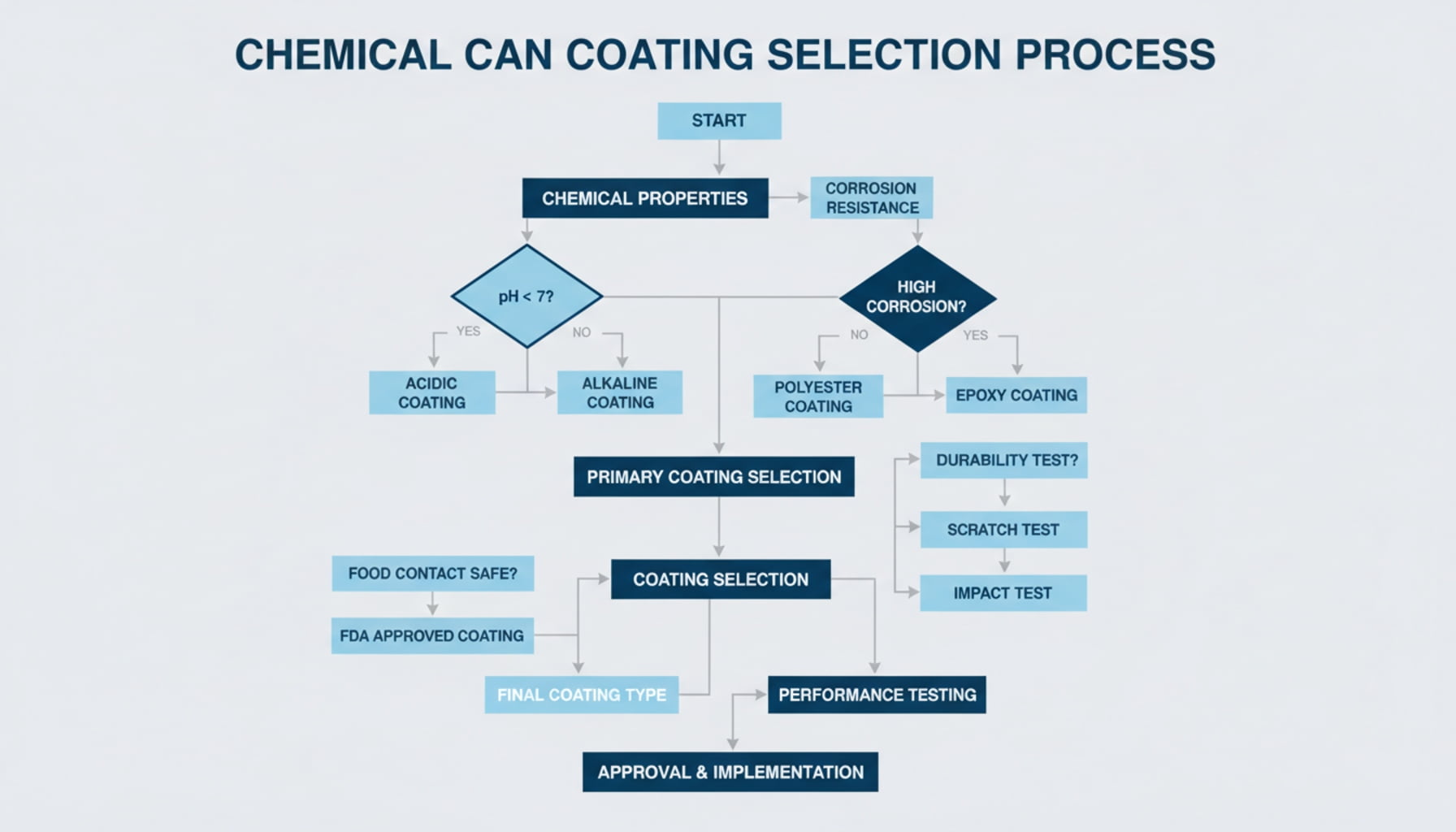
You don't need to feel overwhelmed by all the options. In practice, we follow a very clear and logical path to get to the right answer. It’s a simple framework that forces us to rely on data, not on habits or guesses. Here is the exact process laid out:
- Identify Chemical Properties16: We start by gathering all the data on the product: pH, polarity, key ingredients, etc.
- Assess Risk Level: We ask the first big question: Is this a high-risk chemical (strong acid, oxidizer, etc.)?
- High-Risk Path17: If the answer is yes, the choice is clear. We specify a PVF coating for maximum safety and move to testing.
- Standard-Risk Path: If the answer is no, we move to the next question: Is it water-based or solvent-based?
- Match Coating Type: Based on that answer, we select a preliminary coating—one focused on corrosion resistance (for water-based) or solvent resistance (for oil/solvent-based).
- Evaluate Forming Process: We consider the physical shape of the can. If it requires deep drawing, we ensure the selected coating is flexible enough.
- Mandatory Compatibility Testing18: Finally, with a strong candidate coating chosen, we conduct physical tests with the actual product to confirm our choice.
This structured process ensures that the final recommendation is based on solid evidence, giving you confidence that the packaging is right for your product.
Internal coating selection should never be separated from the packaging material itself.
To understand why metal containers provide better protection for high-risk chemicals, read: Tinplate vs Plastic for Chemical Packaging
When Is Metal Packaging Strongly Recommended Over Plastic?
Plastic containers are sometimes cheaper, and you're considering them to save on costs. But is that saving worth the risk to your product's stability and your supply chain's integrity?
For medium-to-high risk chemicals, products requiring long-term storage, or any goods destined for export, metal packaging with a proper internal coating is strongly recommended. Metal provides superior barrier performance, better mechanical strength, and a lower risk of leakage.

As a manufacturer of metal packaging, you might think I'm biased, but the physical and chemical facts speak for themselves. Plastic packaging certainly has its place, especially for low-risk products with a short shelf life where cost is the primary driver. However, when you are packaging valuable or potentially hazardous chemicals, metal is the superior choice for several key reasons. For a detailed comparison, you can read our in-depth guide:
Tinplate vs Plastic for Chemical PackagingFirst, a coated metal can provides a true hermetic barrier. It is impermeable to gases, light, and moisture. Nothing gets in, and nothing gets out. This is critical for preventing product degradation and for stopping volatile organic compounds (VOCs) from escaping. Second, consider the logistics. Your products will be stacked high on pallets, bounced around in trucks, and shipped across oceans. The mechanical strength and rigidity of steel prevent the crushing, puncturing, and stress-cracking that can affect plastic containers during transport. For any product facing a long or rough journey, the robustness of metal provides a level of security that plastic simply cannot match.
Conclusion
Choosing the right internal coating is a system-level safety decision, not a simple purchase. Preventing just one leakage incident will save your company far more than years of cost-cutting on packaging.
A professional coating decision is a risk-management strategy, not a material choice.
Understanding chemical compatibility is essential to prevent corrosion and leakage, ensuring product safety and stability. ↩
Preventing corrosion and leakage is vital for maintaining product integrity and avoiding costly failures. ↩
Epoxy-phenolic lined cans offer reliable protection against chemical reactions, ensuring long-term product safety. ↩
A chemical containment system ensures the safe interaction between the product and its packaging, preventing contamination. ↩
Knowing chemical reactivity helps in selecting the right coating to prevent adverse reactions and ensure product safety. ↩
Aggressive ions can cause localized corrosion, making it crucial to select coatings that resist such attacks. ↩
PVF coatings offer high chemical inertness, making them ideal for packaging aggressive chemicals safely. ↩
Corrosion resistance is crucial to prevent metal dissolution and ensure the longevity of water-based chemical packaging. ↩
Solvent resistance prevents the breakdown of coatings, ensuring the integrity of solvent-based product packaging. ↩
The pH level determines the type of coating needed to resist acidic or alkaline attacks, ensuring product safety. ↩
Mechanical flexibility prevents coating failure during manufacturing, ensuring the integrity of chemical packaging. ↩
Deep drawing can stress coatings, requiring flexible options to prevent cracking and ensure product safety. ↩
Organosol coatings offer flexibility and chemical resistance, ideal for complex can manufacturing processes. ↩
Compatibility testing validates coating choices, preventing unexpected reactions and ensuring long-term product stability。 ↩
Understanding structured decision logic can help you make informed choices in coating selection, ensuring reliability and safety in your packaging. ↩
Learning how to identify chemical properties is crucial for selecting the right coating, ensuring compatibility and effectiveness in protecting your product. ↩
Exploring the high-risk path can guide you in choosing the safest coating options for hazardous chemicals, minimizing risks and ensuring safety. ↩
Understanding the importance of compatibility testing ensures that the chosen coating will perform well with your product, preventing costly failures. ↩
